AN OUTBREAK HAS BEEN TRACED TO A CONTAMINATED endoscope at Hospital A.” “Several patients were notified that a contaminated flexible endoscope may have been used for their endoscopy procedures at Clinic B.” Headlines like these are becoming the internet to find information regarding incidents where contaminated (unsafe) endoscopes have been used in patient procedures. Several of these situations have been reported in the news media. The Centers for Disease Control and Prevention (CDC) tracks endoscope outbreaks. The US Food and Drug Administration (FDA) also monitors issues associated with flexible endoscopes.
Contaminated flexible endoscopes pose a significant risk to patients. Bacteria can be transmitted from patient to patient, and other sources of contamination may cause infection as well. Biofilm creates a matrix where bacteria are protected so they may grow. In some cases, multi-drug-resistant microorganisms may infect a patient. Those microorganisms are resistant to current medications and may be life threatening to patients. The responsibility for cleaning rests with endoscope reprocessing technicians. Knowledge, training, access to the right tools, and attention to detail are required to make an endoscope safe for patient use.
OBJECTIVE 1: REVIEW THE BASIC CONFIGURATION OF FLEXIBLE ENDOSCOPES
Successful cleaning of a flexible endoscope does not happen by accident. The cleaning process begins with understanding the complexity of the device being cleaned. At first glance, a flexible endoscope looks like a simple device; however, closer inspection proves that to be incorrect. Flexible endoscopes are designed to perform a variety of functions, and each function requires a special component of the endoscope. Each flexible endoscope has a control mechanism that allows the endoscope to be flexed up and down and from side to side during use. This function enables the physician to visualize structures and move the endoscope to improve access during the procedure. Light guides project light through the endoscope and objective lenses enable visualization.
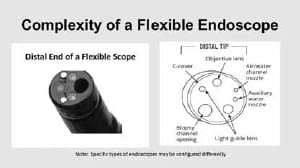
In some endoscopes, channels provide air, water and suction. A biopsy channel allows an instrument to be passed through the endoscope. Each of those functions requires intricate interior design of the device, and that complex design complicates the cleaning process. In addition to a complex design, endoscope channels cannot be visualized with the naked eye, so technicians are challenged to clean areas they cannot see. The distal tip of an endoscope indicates its complexity. Each channel within the endoscope serves a function and it is critical to the overall function of the endoscope. Technicians must clean all areas that become soiled during use and they must do so with little to no ability to see the channels they are cleaning. Figure 2 provides an illustration of the complex interior design of a flexible endoscope.
FIGURE 1: The complexity of the interior of a flexible endoscope is illustrated in the distal tip. Each function represented in the distal tip provides a glimpse of the complexity of the interior of the endoscope. Note: Not all endoscopes are alike. Distal tip and interior configurations vary with the intended function of the endoscope.
OBJECTIVE 2: IDENTIFY CHALLENGES TO CLEANING FLEXIBLE ENDOSCOPES
In addition to their complex configuration, there are other factors that create cleaning challenges when reprocessing flexible endoscopes. The manufacturer’s instructions for use (IFU) must be followed exactly as they are written. When cleaning steps are skipped or when they are not performed for the correct amount of time or with the correct cleaning tools, cleaning errors also occur. This is particularly challenging because all endoscopes require multi-step cleaning. Some flexible endoscopes have over 100 steps in their cleaning process. The manufacturer’s IFU must be readily available and followed to reduce the risk of cleaning errors. The cleaning process can also fail if soil is allowed to remain on the endoscope after its use. When soiled endoscopes are not immediately cleaned, soil may dry, thereby, making the endoscope more difficult to clean. Prolonged exposure
to soil and moisture after use can also enable biofilm to form. Biofilm is a collection of microorganisms that attach to surfaces and each other to form a colony; that colony produces a protective gel that is very difficult for detergents and disinfectants to penetrate. Biofilm in a flexible endoscope is very difficult to remove and poses a danger to patients. All flexible endoscopes should be reprocessed as soon as possible after use to reduce the risk of dried soil and biofilm formation. Some flexible endoscope manufacturers require special cleaning processes (called delayed reprocessing procedures) to be enacted for endoscopes that sit for an hour or longer after they are used. It is important to check the IFU to determine cleaning time restrictions. Having access to the proper tools is another critical component of endoscope cleaning. Cleaning can fail if brushes of the wrong size are used. Brushes that are too small will not make appropriate contact with channel walls and openings. Brushes that are too large may impede the cleaning process. Cleaning solutions and other tools used for flushing and rinsing must also be available and used according to each manufacturer’s IFU. Training also plays a role in the success of the cleaning process. As previously stated, proper cleaning is a multi-step process that must be performed exactly as outlined in the IFU. Endoscope reprocessing technicians must also understand the chemicals and tools used in the cleaning process and must apply that knowledge correctly for every endoscope. No one should reprocess a flexible endoscope alone unless they have had training and have completed a competency evaluation. Time is a significant factor in successful endoscope cleaning. In many cases, endoscopes must be reprocessed and returned to the user department under tight timelines. Pressure is sometimes exerted to move the endoscope through the process quickly. When those timelines are unrealistic and demands for turnaround are forceful, technicians may feel tempted to skip steps or cut corners in the cleaning process; this can lead to unsafe endoscopes being released for use. In some cases, the cleaning process may be successful, but the endoscope may become recontaminated after cleaning; this may occur due to endoscope mishandling (e.g., handling a clean endoscope while wearing soiled PPE or placing an endoscope on an unclean surface after cleaning). Moisture is another danger for endoscope contamination. If a flexible endoscope is not completely dried, any moisture that remains in the endoscope’s channels creates an environment where bacteria can grow and multiply. Bacteria multiply quickly and as they do, the level of contamination of an endoscope grows as well. Figure 3 provides an example of how quickly bacteria can grow. When an endoscope containing moisture is stored, its dark, wet lumens and crevices provide a receptive place for microorganisms to multiply.
FIGURE 3: When the endoscope is removed from the storage cabinet and placed into service, it is contaminated and poses a threat to patient safety.
OBJECTIVE 3: DISCUSS THE PREVALENCE OF DIRTY FLEXIBLE ENDOSCOPES
Even if dirty endoscopes were a rare occurrence in a healthcare facility, their occurrence would still be unacceptable. Unfortunately, incidents involving dirty endoscopes are all too common. In its 2018 Top 10 List of Health Technology Hazards, ECRI Institute cited endoscope reprocessing failures and improper cleaning as the number 2 and number 5 patient risks, respectively. A study conducted by Ofstead and Associates revealed that 99% of the time, one or more reprocessing steps were skipped or completed incorrectly. Multiple steps were skipped 45% of the time. Every endoscope reprocessing area should audit their cleaning processes to help ensure each step is being performed exactly as stated in the device manufacturer’s IFU. Dirty endoscopes pose a significant risk to patients. There is a misconception that a dirty endoscope can be made safe by a high-level disinfection or sterilization process. It is important to remember that soil and bacteria remaining in an endoscope will not be eradicated by disinfection or sterilization processes. Those processes cannot compensate for inadequate cleaning. Put simply, any endoscope that is not clean will remain dirty and dangerous when used on the next patient. Each facility and technician must evaluate practices and take necessary steps to reduce the incidence of dirty flexible endoscopes. Dirty flexible endoscopes are a patient safety issue and their elimination should be prioritized.
CONCLUSION
Whenever a dirty endoscope is used in a procedure, the patient is put at risk. Several factors have an impact on the process of making a flexible endoscope safe for patient use; this includes the device’s configuration, complex cleaning instructions, and human factors. Dirty endoscopes put patients at risk of infection. Training, proper cleaning tools and meticulous attention to detail can reduce that risk.
RESOURCES
International Association of Healthcare Central Service Materiel Management. 2017. Endoscope Reprocessing Manual, First Edition. Ofstead C, et al. Re-evaluating Endoscopy- associated Infection Risk Estimates & their Implication. American Journal of Infection Control. 41 (2013), pp. 734-6. 2. Ofstead C, et al. Endoscope Reprocessing Methods: a Prospective study on the Impact of Human Factors and automation. Gastroenterology Nursing, July/Aug 2010. ECRI Institute. 2018 Top 10 Health Technology Hazards. https://www.ecri.org/Resources/Whitepapers_and_reports/Haz_18.pdf Association for the Advancement of Medical Instrumentation. ANSI/AAMI ST91:2015. Flexible and semi-rigid endoscope processing in health care facilities.
Supervisory Continuing Education (SCE) lessons provide members with ongoing education focusing on supervisory or management issues. These lessons are designed for CER re-certification, but can be of value to any CRCST in a management or supervisory role. Earn Continuing Education Credits: Online: Visit www.iahcsmm.org for online grading at a nominal fee. By mail: Mailed submissions to IAHCSMM will not be graded and will not be granted a point value (paper/pencil grading of the SCE Lesson Plans is not available through IAHCSMM or Purdue Univer-sity; IAHCSMM accepts only online subscriptions). Scoring: Each online quiz with a passing score of 70% or higher is worth two points (2 contact hours) toward your CER re-certification (6 points) or CRCST re-certification (12 points). More information: IAHCSMM provides online grading service for any of the Lesson Plan varie-ties. Purdue University provides grading services solely for CRCST and CIS lessons.