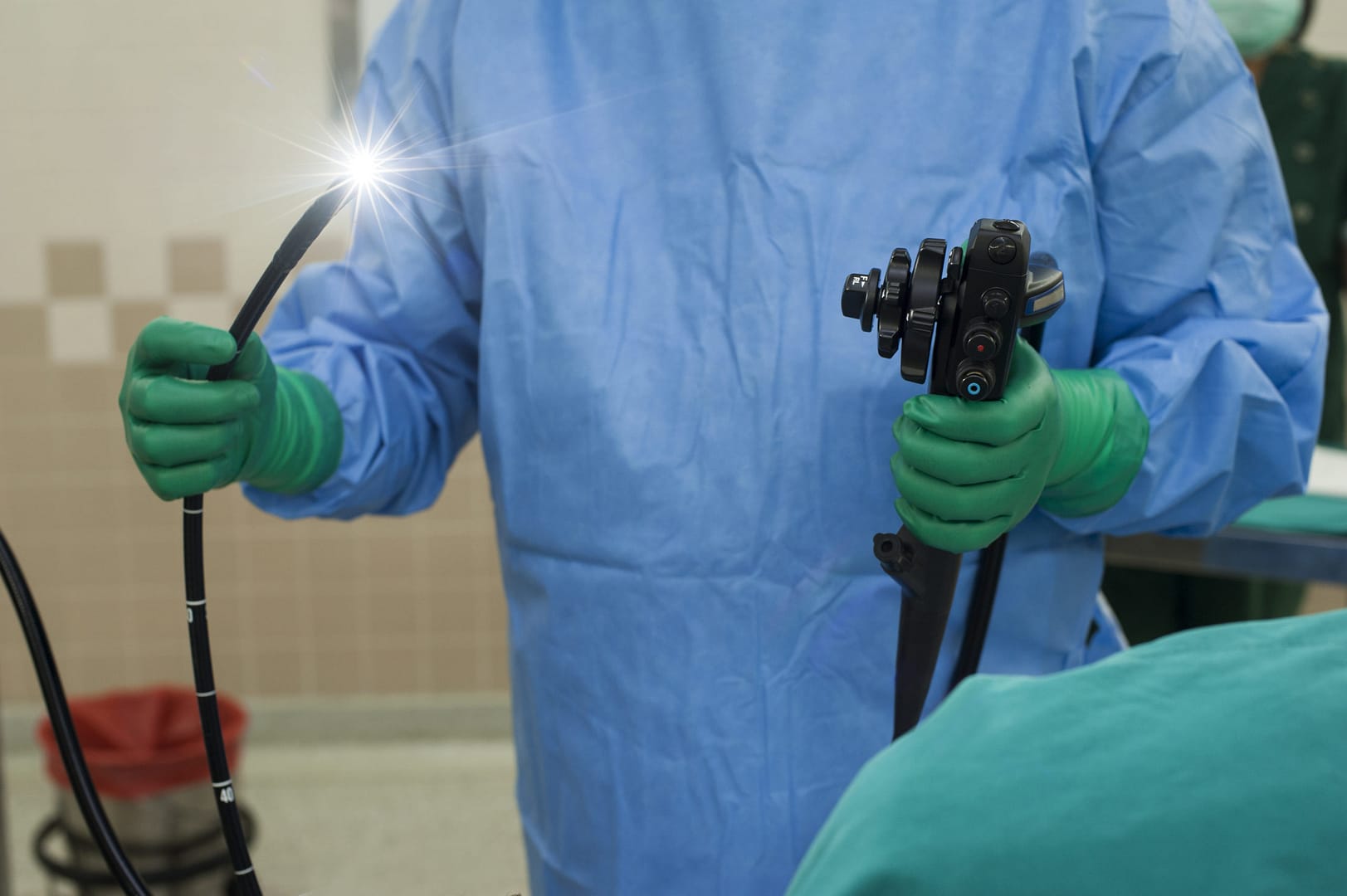
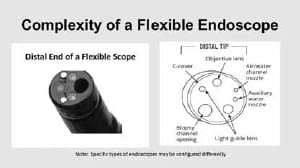
The Sterile Processing Department (SPD), also known as the Central Sterile Services Department (CSSD), is the area in a hospital where cleaning and sterilization of devices used in medical procedures takes place. The processes an instrument goes through in the CSSD depends on its use, material construction, and other factors. Typically, an instrument coming from the Operating Room arrives at the Decontamination area to be manually cleaned, then placed in an ultrasonic cleaner or sonic irrigator before being placed into a washer/disinfector. Next, the instrument would be transported to a Prep & Pack area to be prepared for sterilization. It will then be sterilized through one of many types of sterilization processes. Once sterilized, the instrument will either be sent back to a procedure room to be used again, or into sterile storage until it is needed again for a procedure. Sterile Processing Department Technicians are required to wear Personal Protective Equipment (PPE) as a safety precaution to prevent exposure to potentially infectious bacteria.
1 THE DECONTAMINATION PROCESS
Soiled instruments from the OR are first taken to the decontamination area to be cleaned of gross soils and inspected for damage. The decontamination process is an important stage in device reprocessing because an instrument or device cannot be sterilized until fully clean. In the decontamination process, soiled instruments are sorted, inspected and if necessary, disassembled. Instruments are first manually cleaned. Depending on the device, they may then go through an automated washing process following manual cleaning. The reasons CSSD staff may choose to manually clean a device/instrument include: The OEM recommends the device be manually cleaned Hospital policy dictates the device must be manually cleaned and then processed in an automated washer/disinfector
Delicate instruments or powered equipment may not be suitable to be processed in an automated washer/disinfector
The hospital may not have a washer/disinfector
Manual cleaning requires either a two-bay sink or three-bay sink. In a three-sink method, each bay plays a role in the cleaning process.1 Sink 1: Instruments are immersed in an enzymatic solution to begin breaking down soils
Sink 2: Instruments are immersed in a detergent solution and manually brushed
Sink 3: Instruments are thoroughly rinsed with clean, treated water
If a two-bay sink is being used, the process combines the enzymatic solution and detergent solution in one bay. The second bay contains clean, treated water as with the three-bay sink. If cleaning a lum
ened instrument or device, a brush or flushing with pressurized water may be used to loosen soils. Lubricant may be applied after manual cleaning. Explore our Decontamination Sinks
The reasons CSSD staff may choose to mechanically clean a device using an ultrasonic cleaner or irrigator and a washer/disinfector include: Washer/disinfectors offer increased productivity compared to staff manually cleaning Washer/disinfectors provide a consistent, repeatable cleaning process so staff can be sure devices are thoroughly cleaned every time
One form of mechanical cleaning is ultrasonic cleaning. Ultrasonic cleaners clean instruments through acoustic cavitation, which forms air bubbles that implode on an instrument’s surface. These air bubbles can reach small crevices and hard-to-reach areas on a device. Ultrasonic cleaners are typically used to clean devices that may be sensitive to damage, and are too delicate for a traditional washer/disinfector. Ultrasonic cleaners have two chambers, and may come in a variety of sizes and types depending on the department’s need: freestanding, tabletop, large capacity, etc.
Explore our Ultrasonic Cleaners
The mechanical cleaning process may also be done via automated washer/disinfectors, which are available as single-chamber or multi-chamber. The washer/disinfector combines impingement, water temperature, and detergent to clean devices.
Explore our Washer/Disinfectors In both ultrasonic cleaner and washer/disinfector processes, cleaning indicators are often used to monitor and evaluate the performance of the wash cycle.
THE STERILIZATION PROCESS
Once the instrument has been manually cleaned, mechanically cleaned, or both, it will be sent to the preparation and packaging area of the SPD. Once the instrument pack has been prepped for sterilization, it is ready to be sterilized through one of many methods of sterilization. The main methods of medical instrument sterilization include:1 Steam Sterilization – Steam sterilization is the predominant form of sterilization in SPDs. A steam sterilizer, also known as an autoclave, is suitable for sterilizing heat and moisture-stable items. Steam sterilization cycle types include gravity, pre-vacuum and SFPP (Steam Flush Pressure Pulse). Cycle time varies according to cycle type, load weight and density and other variables such as exposure and drying time. At the end of the sterilization cycle, the SPD technician reviews the sterilizer printout to verify if all sterilization parameters have been met.
Biological and chemical indicators are used to monitor the sterilization process and indicate if the load was exposed to the appropriate conditions to achieve sterility. Explore our Steam Sterilizers Low Temperature Sterilization – Other forms of sterilization may include low temperature sterilization methods like ethylene oxide (EtO), vaporized hydrogen peroxide, liquid chemical and ozone. Ethylene Oxide Sterilization consists of five stages – preconditioning and humidification, gas introduction, exposure, evacuation, and air washes – and takes about 1 – 4.5 hours. Aeration time makes the cycle time longer – an additional 12 – 36 hours.
Vaporized Hydrogen Peroxide Sterilization, Also known as hydrogen peroxide gas sterilization, is a low temperature sterilization process commonly used to sterilize heat-sensitive devices. A hydrogen peroxide sterilization process involves H2O2 vapor filling the sterilization chamber, contacting and sterilizing exposed device surfaces. Once the sterilization cycle has completed, the vapor is removed from the chamber and converted to water and oxygen.
Liquid Chemical Sterilization provides safe and effective reprocessing of heat-sensitive, critical and semi-critical devices such as duodenoscopes and other complex endoscopes.
Ozone Sterilization is one of the newest of the sterilization methods. With an extra oxygen atom attached to the molecule, ozone is able to destroy microbes to sterilize devices. Ozone leaves no chemical residue and there are no toxic emissions.
STERILITY ASSURANCE Ensuring that an instrument is sterile and safe to use is vital to the reprocessing cycle. Sterility assurance monitoring can be done through various forms of test packs, chosen based on the type of sterilization process used or parameters being measured. A passing biological and chemical indicator test confirms that specific parameters of a sterilization cycle were met. Some types of sterility assurance products include:
1 Biological Indicators – Biological indicators (BI) are designed to challenge the lethality of a sterilization process to kill bacterial spores. Biological indicators are used within process challenge devices or challenge packs for routine monitoring, load monitoring and qualification of the sterilization systems. The frequency for using biological indicators is based on the standards, the manufacturer’s instructions for use and the facility policies and procedures. A passing BI result indicates that the load can safely move on to sterile storage or the OR. Chemical Indicators – Chemical Indicators (CI) may be applied externally or internally to the package container. External chemical indicators, also called process indicators, show that the set has been fully exposed to the sterilization process. Internal chemical indicators, which come in a variety of forms, are placed in the most challenging area of the set and are read by OR staff to confirm that sterilant penetrated the load.
Bowie Dick Test – Bowie Dick Tests are required for steam sterilizers with a pre-vacuum cycle to check the efficiency of the air removal and steam penetration in the chamber. This test must be run daily before any pre-vacuum cycles are run.